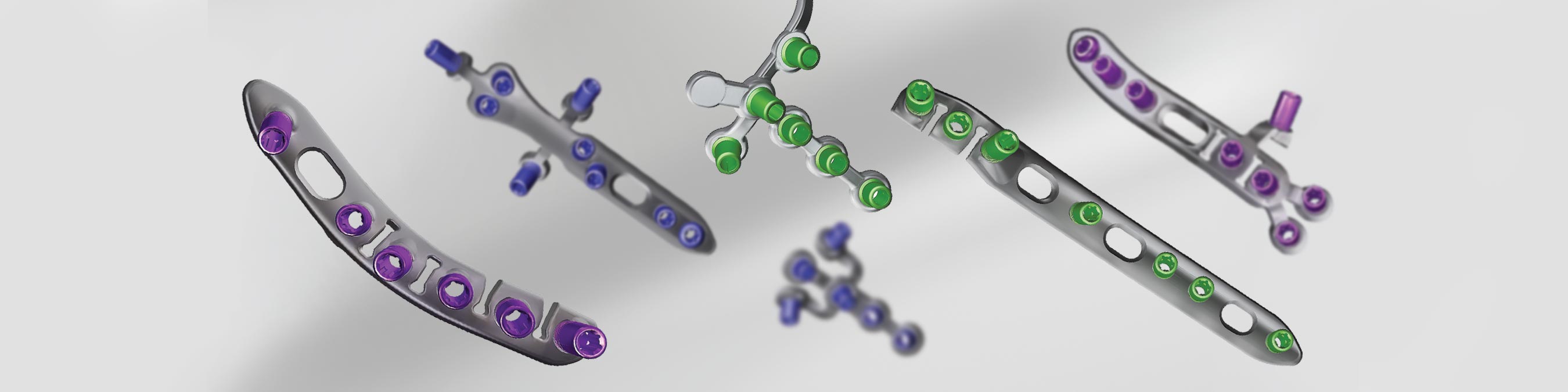
The A.L.P.S. ® Elbow Fracture System offers an extensive selection of anatomically contoured implants to address fractures around the elbow. The system includes medial, posterior lateral, olecranon, coronoid and proximal radial plates. Each plate is designed to match the anatomy at the specified location. Additional contouring can be performed in situ for fine adjustments.
Multi-directional locking screw technology provides versatility and strength. With a cone of angulation of up to 25̊, the screws simplify the targeting and capturing of fragments. The locking screws lock into the plate by creating their own thread.
- Low profile plate design and smooth implant surface to minimize soft tissue irritation
- Bullet tip minimizes soft tissue disruption during insertion
- Engineered from TiMAX® material for strength and biocompatibility
- Preloaded F.A.S.T. Guide® inserts enable percutaneous application with fast, accurate drilling
- F.A.S.T. Tabs® technology allows in situ contouring for true plate-to-bone conformity
- Available in small and large sizes to assist in matching the anatomy
- Adapters available for fixed-angle K-wire placement for provisional fixation
- Conversion washers available for use with 3.5mm non-locking screws